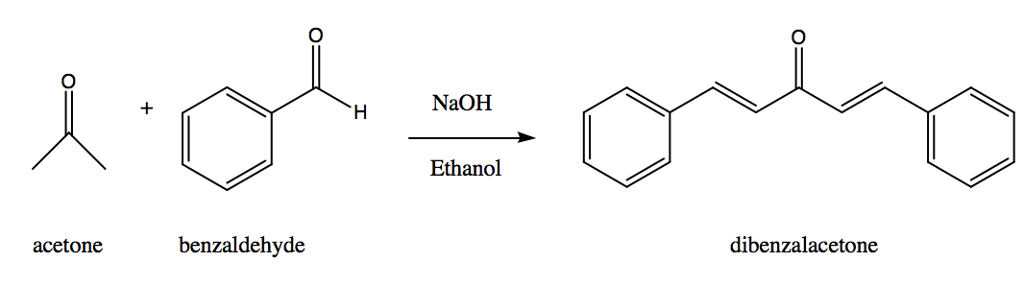
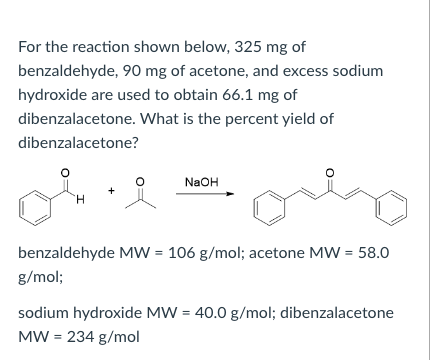
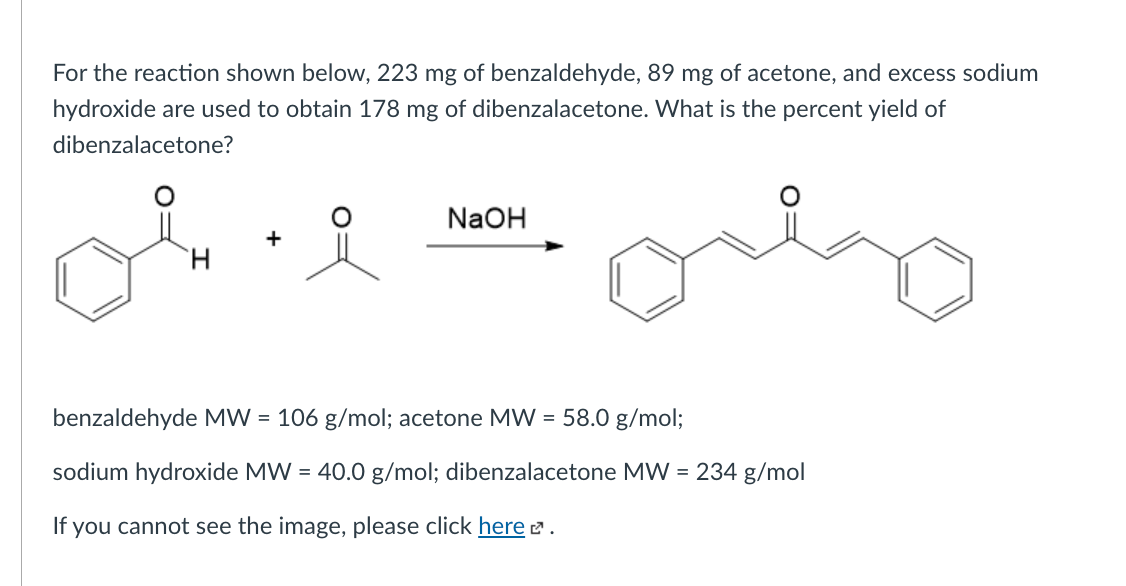
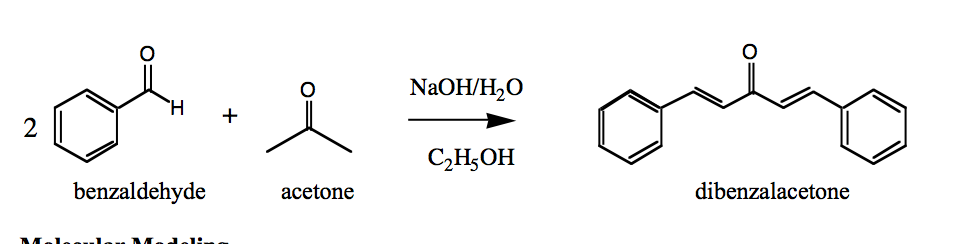
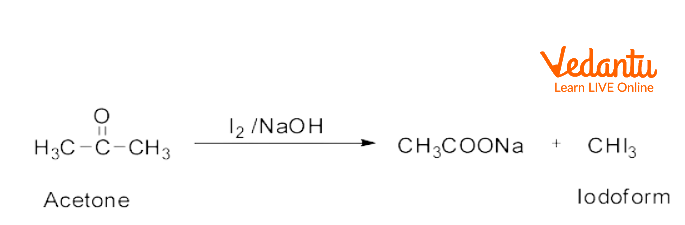SOLVED: 2 Why are you adding the mixture of acetone benzaldehyde dropwise to the sodium hydroxide solution? Why not add the acetone to the sodium hydroxide solution and then add the benzaldehyde
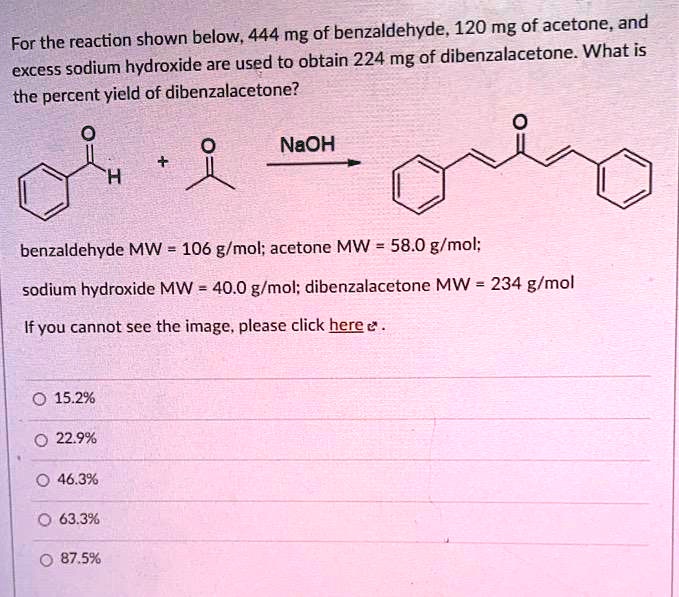
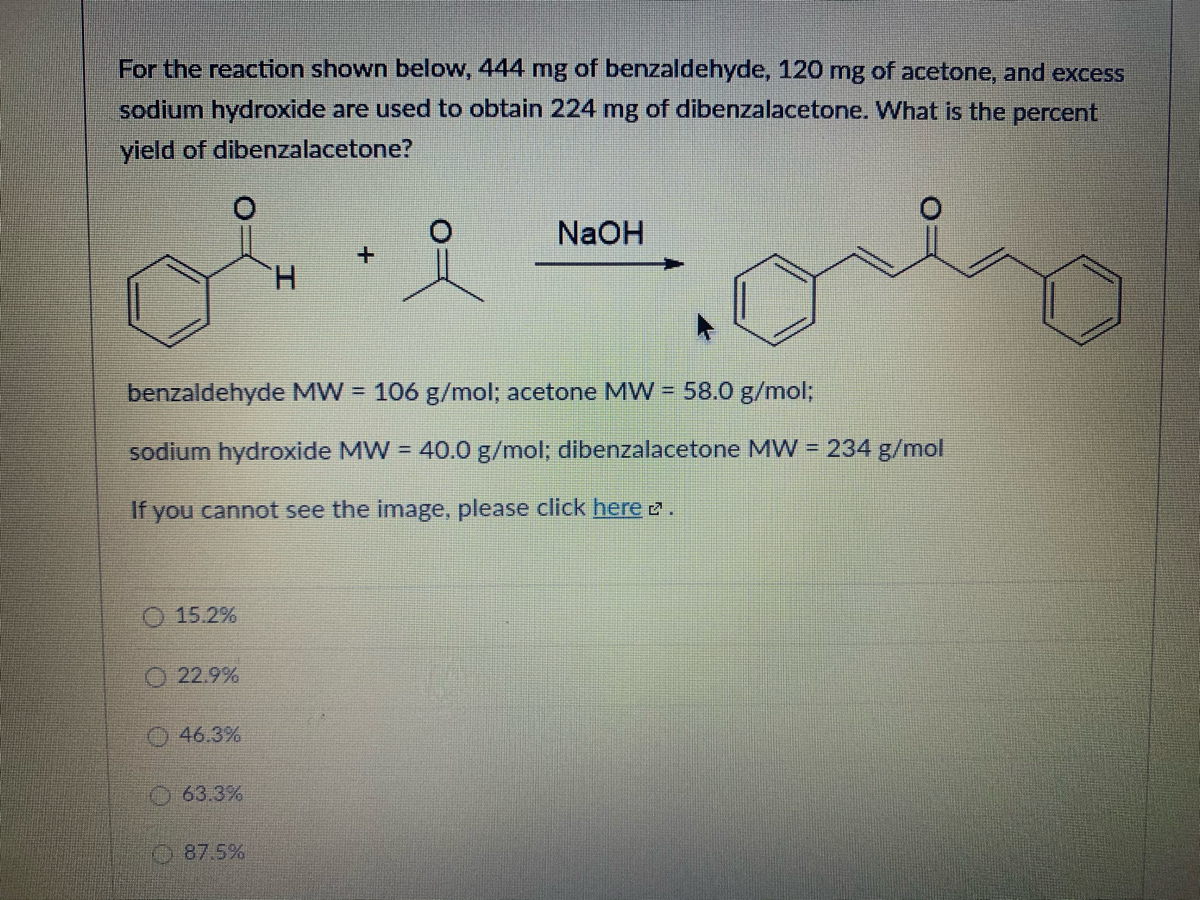
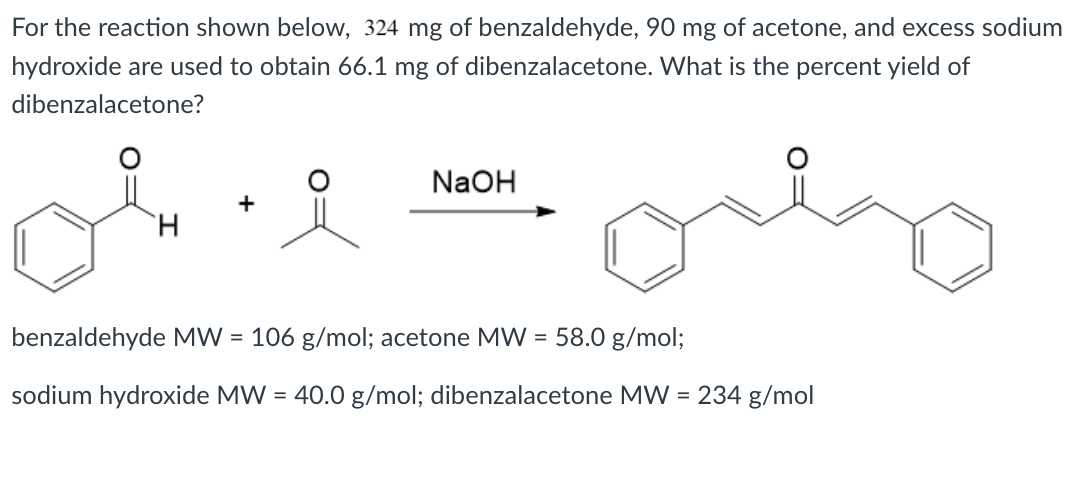
SOLVED: 444 mg of benzaldehyde; 120 mg of acetone and for the reaction, excess sodium hydroxide are used to obtain 224 mg of dibenzalacetone. What is the percent yield of dibenzalacetone? NaOH
organic chemistry - Color changes of a solution of acetone and sodium hydroxide - Chemistry Stack Exchange

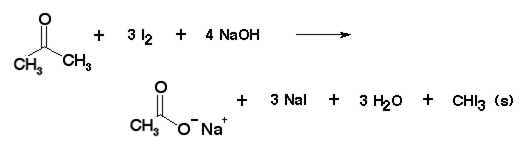
SOLVED: Ldoorm test (Ydeo Z) a) Write the reaction of acetone with iodine in the presence of sodium hydroxide: b) Based on Video 2, how can you know the reaction was positive?