
OneClass: A standard entropy of vaporization of acetone is approximately 85jk/mol at its boiling poin...

SOLVED: Calculate the change in entropy that occurs in the system when 1.97 mol of acetone (C3H6O) condenses from a gas to a liquid at its normal boiling point (56.1 °C). The
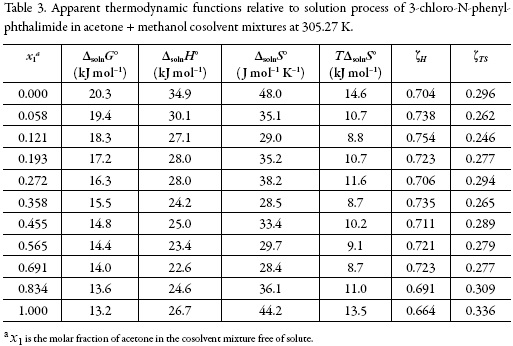
Solution thermodynamics and preferential solvation of 3-chloro-N-phenyl-phthalimide in acetone + methanol mixtures

SOLVED:The molar heat of vaporization of acetone, C3 H6 O, is 30.3 kJ / mol at its boiling point. How many kilojoules of heat would be liberated by the condensation of 5.00

Heat of vaporization of acetone. Simulation data: • this work, AUA4... | Download Scientific Diagram

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram

SOLVED: If the enthalpy of vaporization of acetone is 32.0 kJ/mol, what mass of acetone (molar mass equals 58.08 g/mol) will be vaporized when 121 kJ of energy (as heat) are added

SOLVED: The standard heat of vaporization of acetone is 31.3 kJ/mol and its normal boiling point is 56°C. Calculate the standard entropy of vaporization. 10.5 J/mol K 0.095 J/mol K 559 J/mol




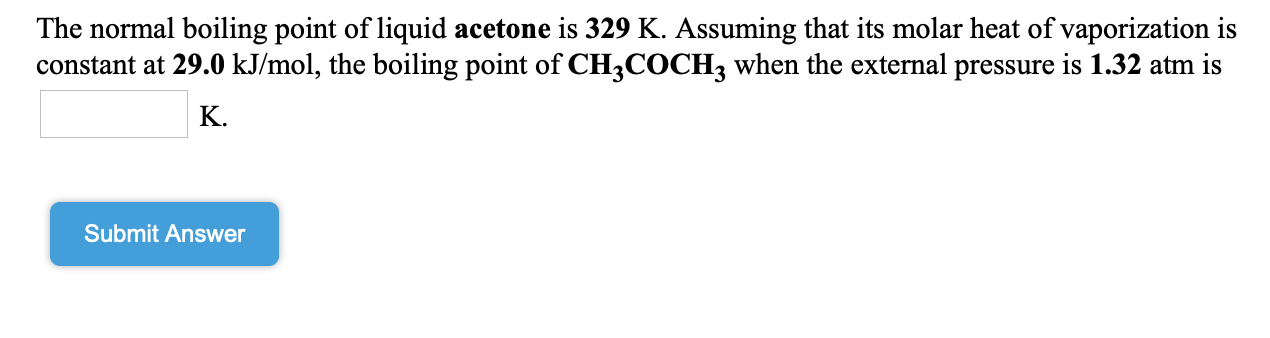
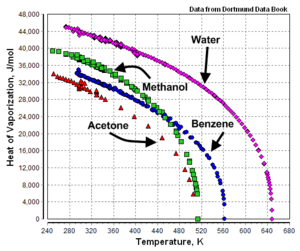



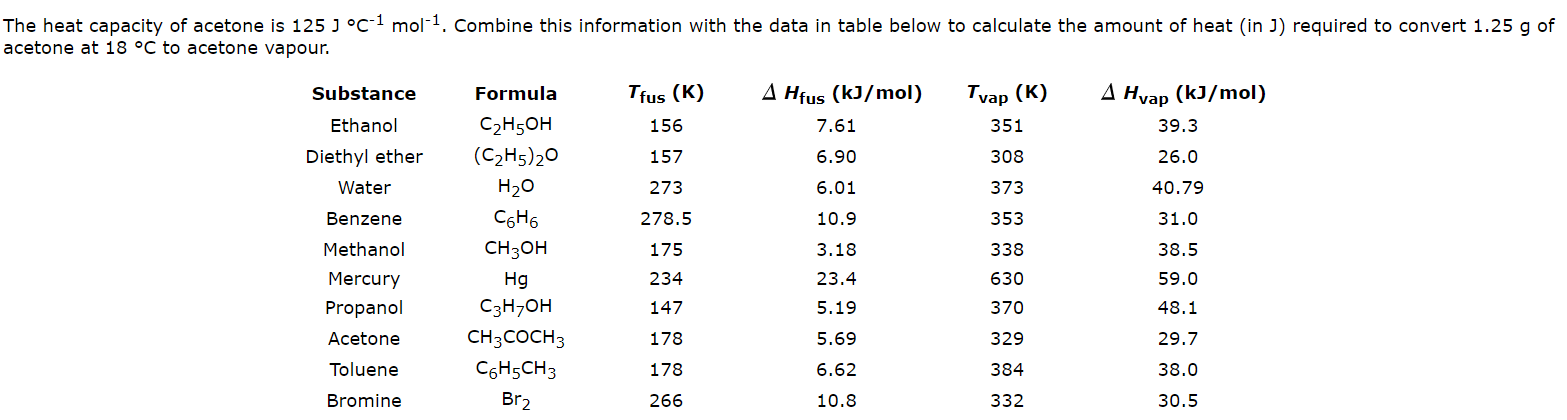
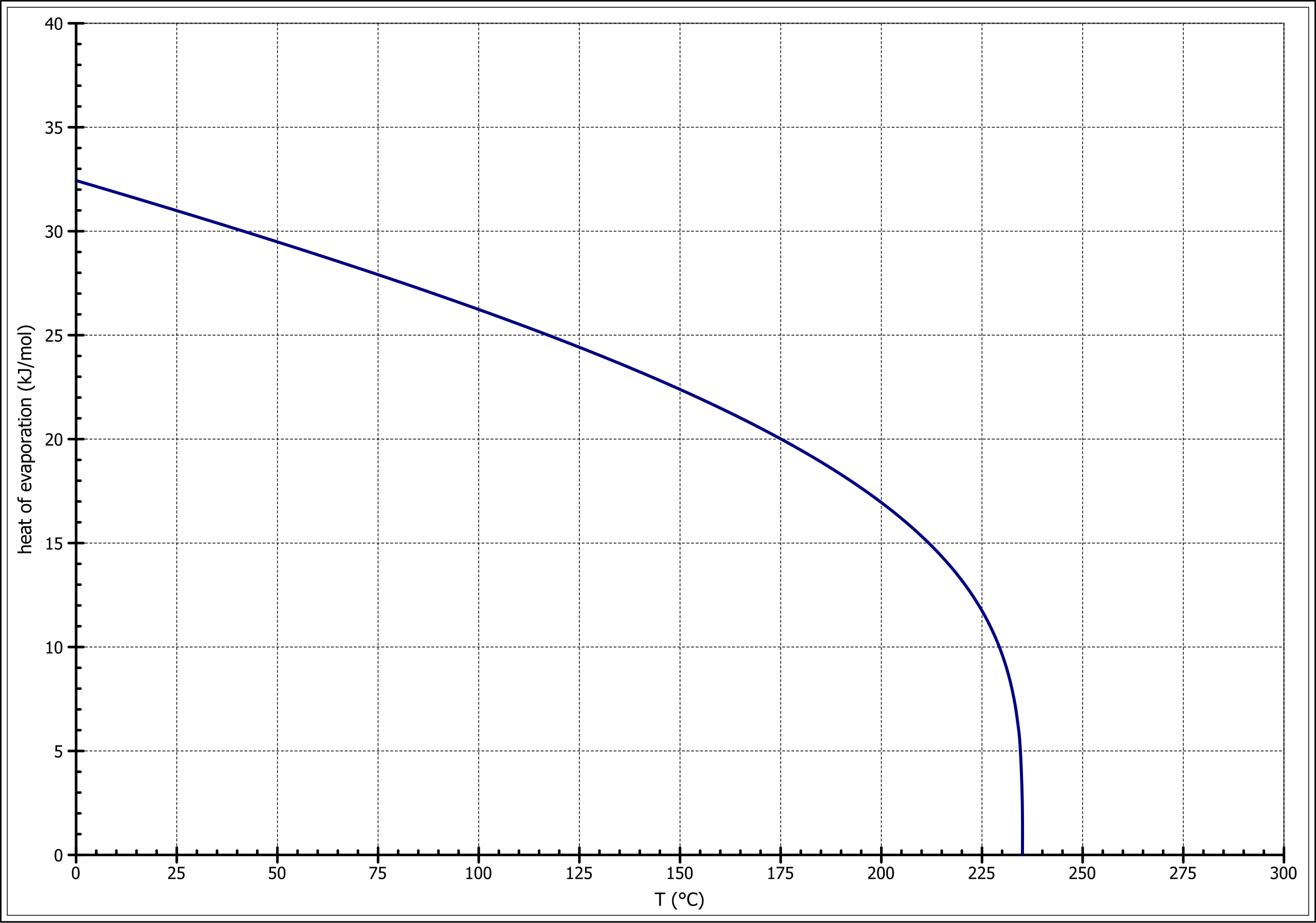

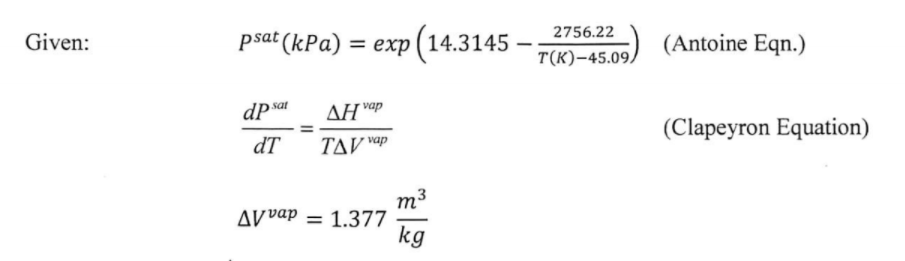
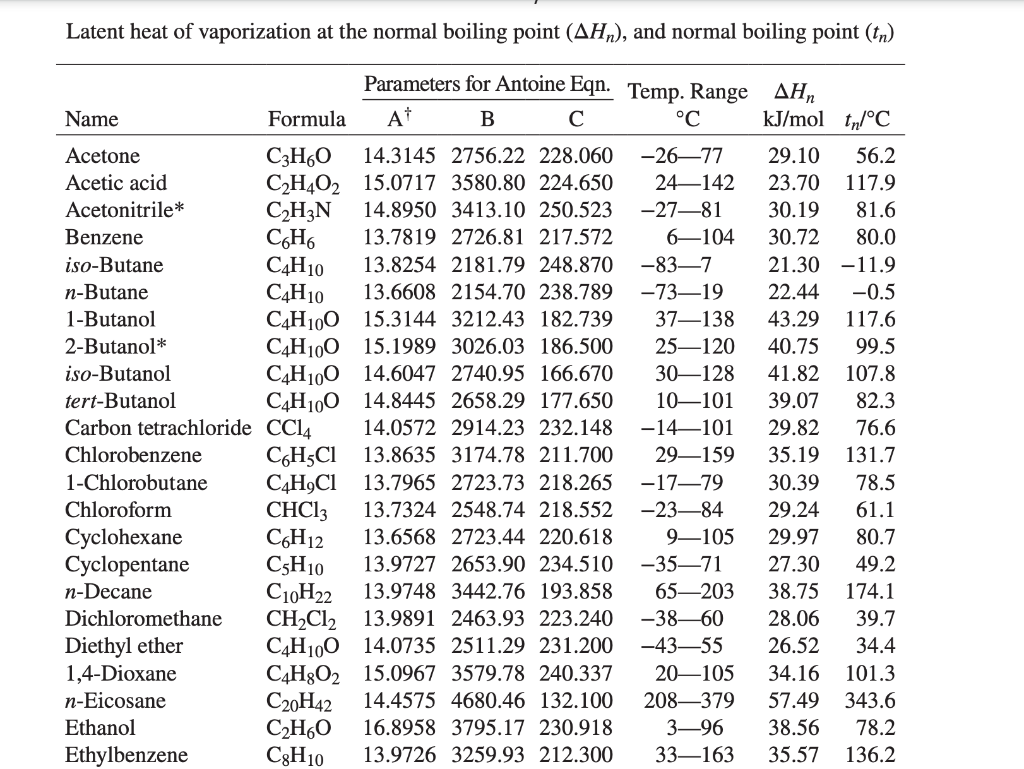

![Solved] Estimate the heat of vaporization (kJ/mol | SolutionInn Solved] Estimate the heat of vaporization (kJ/mol | SolutionInn](https://s3.amazonaws.com/si.question.images/images/question_images/1590/0/8/2/6645ec6bc68ba9271590082649906.jpg)