
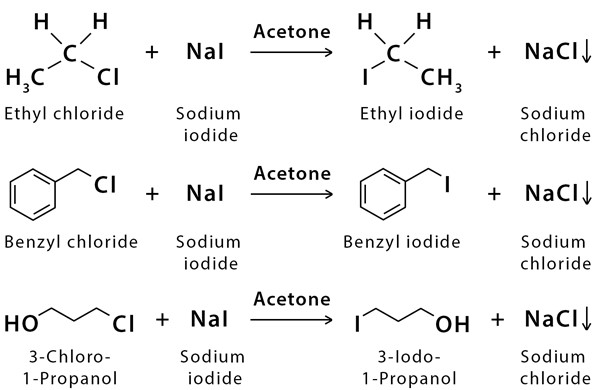
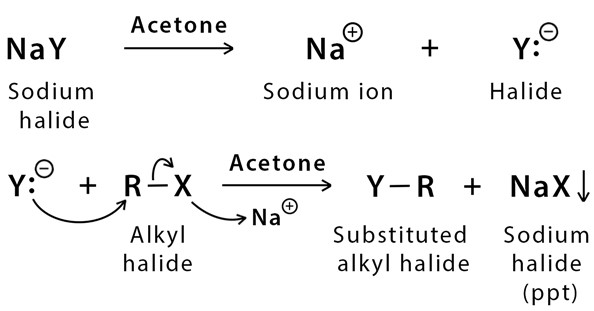
SOLVED: 1. Sodium iodide in acetone: Nal readily dissolves in acetone: The moderately polar aprotic solvent promotes Sn? reactions lodide is an excellent nucleophile: The solvent does not favor ionization of alkyl

Solubility, Density and Solution Thermodynamics of NaI in Different Pure Solvents and Binary Mixtures | Journal of Chemical & Engineering Data

PDF) Solubility of Sodium Halides in Aqueous Mixtures with ε-Increasing Cosolvents: Formamide, N -Methylformamide, and N -Methylacetamide at 298.15 K
Thermodynamics Studies on the Solubility of Inorganic Salt in Organic Solvents: Application to KI in Organic Solvents and Water

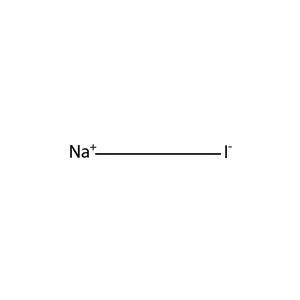
Statement - 1 : CH3 - CH2 - Cl + NaI Acetone→ CH3 - CH2 - I + NaCl Statement - 2 : Acetone is polar - protic solvent and solubility order
What is an equation, clearly showing the stereochemistry of the starting material and the product, for the reaction of 2-bromobutane with sodium iodide in acetone? - Quora









![What is the name of the following reaction ? CH3CH2CH2Br [dry acetone]NaI CH3CH2CH2I What is the name of the following reaction ? CH3CH2CH2Br [dry acetone]NaI CH3CH2CH2I](https://i.ytimg.com/vi/hxgro16PgJg/maxresdefault.jpg)